
Why enzymes are important
Chances are your biochemistry unit is not a student favorite. I often hear “why are we learning chemistry in biology class?” But to really understand how cells work, students need to understand macromolecules. (If you need some biochemistry lesson ideas, check out this blog post). Luckily, there are fun labs you can do to help students understand enzymes.
Using catalase to test enzyme efficiency
A common enzyme lab for students to measure the impact of temperature and pH on the efficiency of catalase. Catalase is an enzyme is found in almost all living organisms that breaks down hydrogen peroxide (H2O2) into oxygen and water. Many teachers use raw chicken liver or potato as the source of the catalase. I’ve done both and frankly potato is less stinky and is easier to clean up after. Here is the gist of the lab:
- Students will need: potato puree, tweezers, a beaker full of hydrogen peroxide, and a stopwatch.
- Peel a raw potato and cut it into pieces. Place the potato in the blender and add a small amount of water. Puree until smooth. (One large potato should be enough for 1 class period).
- Note: The potato will turn brown relatively quickly as it comes in contact with the air. Don’t worry! This does not impact the results of the experiment.
- Collect the paper discs out of your hole puncher (or hit up the copy center at your school).
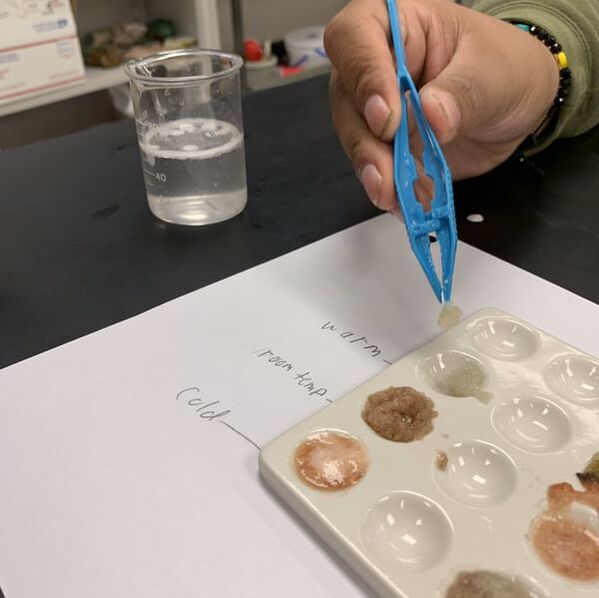
- Using tweezers, have students dip a paper disc in the potato puree. Place the paper in the bottom of the beaker of peroxide and start the stopwatch. As the catalase on the paper disc breaks peroxide into oxygen and water, the disc will float. Have students time how long it takes for the paper to rise.
- Concentration: To show students the impact of concentration on enzyme efficiency, simply water down the potato puree. Do a trial with straight potato, a trial with a 3:1 ratio of potato to water, and a trial with 1:1 ratio of potato to water.
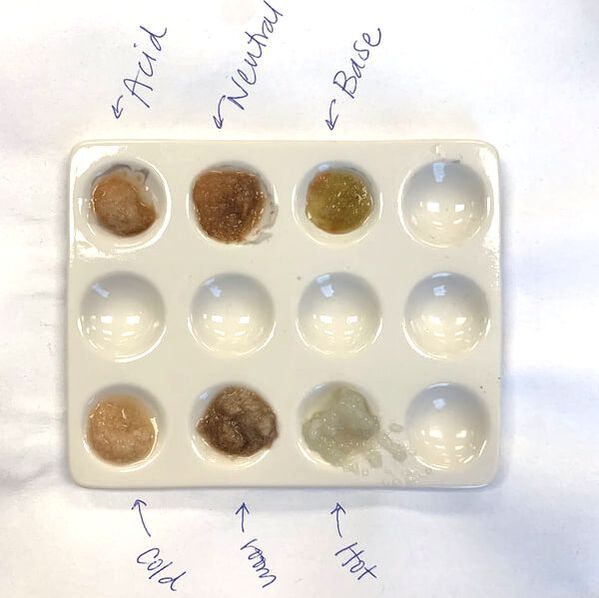
- pH: To show students the impact of pH on enzyme efficiency, have them add a few drops of an acid and a base to the potato purees on a spot plate. Vinegar and bleach are great options. Repeat the experiment and have students determine at which pH catalase works best.
- Temperature: To measure the impact of temperature on enzyme efficiency you have two options.
- Option 1: Change the temperature of the peroxide. Place a beaker of peroxide in an ice bath, and another in a warm water bath. This option tends to yield the best results.
- Option 2: Change the temperature of the potato puree. This can be done easily by putting some of the puree in the fridge and some in the microwave (or boil it at home ahead of time). This does not always give the best results because the cold potato can warm up pretty quickly, but still works if you don’t have water baths available.

Troubleshooting Tips
After doing this lab for multiple years, here are some additional tips:
- Have students do multiple trials (at least 3) and take the average. Sometimes they get weird data, so this helps with accuracy.
- If you are testing multiple variables, have students get fresh peroxide before starting the new variable. For example, have students collect all the temperature data, get fresh peroxide, and then collect pH data.
- If the paper disc takes more than 1 minute to rise, tell students that the enzyme is denatured and they can stop timing and move on to the next trial.
- When I first started doing this lab I used petri dishes for all the potato purees and it was a lot of clean up. I recently switched to chemistry spot plates (pictured above) and it made clean up so much easier!
If you would like to purchase a lab write-up, you can find it here:
Rock on,





